2025
- E. Söderberg, M. R. Molenaar, K. Zaczyka, M. Johansson, M. A. Hayes, P.-O. Syrén.* Engineering of an ancestral McbA with enhanced domain mobility extends biocatalytic amide synthesis scope. Adv. Synth. Catal. 2025, e70232. https://advanced.onlinelibrary.wiley.com/doi/abs/10.1002/adsc.70289

- . Munthe, L. Bergström, D. Bolinius, I. Cotgreave, A.-K. Hellström, H. Holmquist, M. J. Johansson, A. Julander, R. Lihammar, J. Martin, B. Martín-Matute, A. P. Mathew, T. Rydberg, L. Skedung, M. Witala, P.-O. Syrén. Progress and future outlook towards a safe and sustainable production and use of chemicals. Commun. Chem. 2025, 8, 350. https://www.nature.com/articles/s42004-025-01785-8

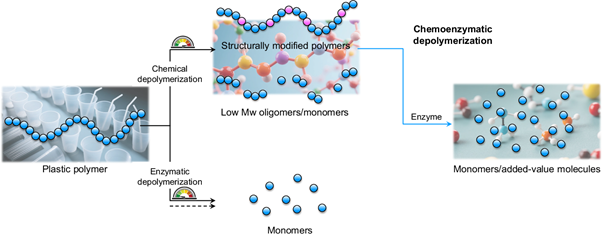
- S. Sun, P.-O. Syrén.* Chemoenzymatic cascade depolymerization of plastics. Commun. Chem. 2025, 8, 272 https://www.nature.com/articles/s42004-025-01679-9

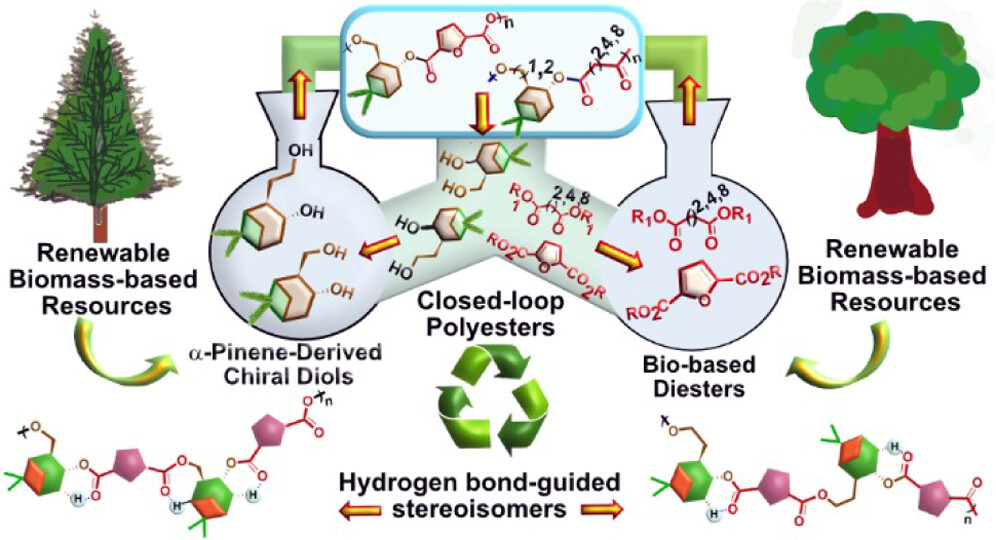
- G. Ranjani, X. Lopez-Lorenzo, S. Subramaniyan, M. Hakkarainen, P.-O. Syrén.* Chemically Recyclable and Enzymatically Degradable Polyesters from α-Pinene-Derived Chiral Diols. ACS Sustain. Chem. Eng. 2025 13, 6696–6705 https://pubs.acs.org/doi/full/10.1021/acssuschemeng.5c01374

- U. Hoffmann, A. Schuppe, A. Knave, E. Sporre, H. Brismar, E. Englund, P.-O. Syrén, Elton P Hudson. A Cyanobacterial Screening Platform for Rubisco Mutant Variants. ACS Synth. Biol. 2025, 14, 2619–2633

- Enzymatic degradation of phthalate esters in the environment: Advances, challenges and opportunities. S. Sun, P.-O. Syrén.* Chem. Eng. J. 2025 508, 160640 https://www.sciencedirect.com/science/article/pii/S1385894725014615


2024 - X. Lopez-Lorenzo, D. Hueting, E. Bosshard,P.-O. Syrén.* Degradation of PET microplastic particles to monomers in human serum by PETase. RSC Faraday Discuss. 2024, 252, 387–402 https://pubs.rsc.org/en/content/articlelanding/2024/fd/d4fd00014e


- X. Lopez-Lorenzo, G. Ranjani, P.-O. Syrén.* Conformational Selection in Enzyme-Catalyzed Depolymerization of Bio-based Polyesters. ChemBioChem 2024, e202400456 https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/cbic.202400456


- S. Subramaniyan, B. Zhang, P.-O. Syrén, M. Hakkarainen. Designing from biobased to closed-loop circularity: Flexible dynamic polyimine-amide networks. Chem. Eng. J. 2024 501, 157632. https://www.sciencedirect.com/science/article/pii/S138589472409123X

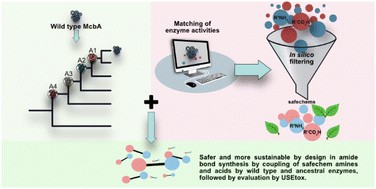
- E. Söderberg, K. Felicitas von Borries, U. Norinder, G. Ranjani, M. Petcheye, S. Chavan, H. Holmquist, M. Johansson, I. Cotgreave, M. Hayes, P. Fantke, P.-O. Syrén*. Toward safer and more sustainable by design biocatalytic amide-bond coupling. Green. Chem. 2024, 26, 11147 – 11163 https://pubs.rsc.org/en/content/articlelanding/2024/gc/d4gc03665d

2023
- D. Hueting, K. Schriever, R. Sun, S. Vlachiotis, F. Zuo, L. Du, H. Persson, C. Hofström, M. Ohlin, K. Walldén, M. Buggert, L. Hammarström, H. Marcotte, Q. Pan-Hammarström, J Andréll, P.-O. Syrén*. Design, structure and plasma binding of ancestral β-CoV scaffold antigens. Nat. Commun. 2023, 14, 6527: doi: 10.1038/s41467-023-42200-x

- B. Guo, X. Lopez-Lorenzo, Y. Fang, E. Bäckström, A. Jose Capezza, S. Reddy Vanga, I. Furó, M. Hakkarainen, P.-O. Syrén*. Fast Depolymerization of PET Bottle Mediated by Microwave Pre-Treatment and An Engineered PETase. ChemSusChem 2023, 16, e202300742. doi: 10.1002/cssc.202300742


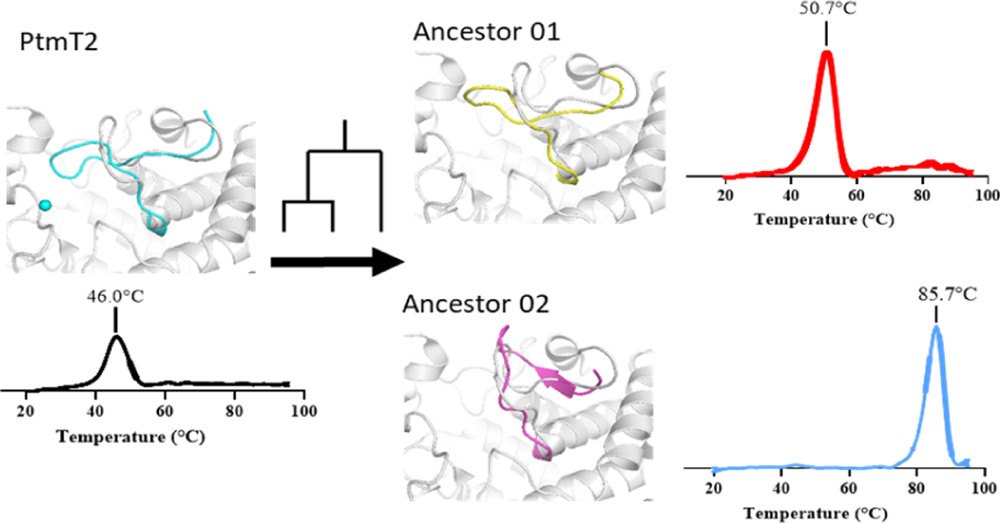
- A. Hueting, S. R. Vanga, P.-O. Syrén*. Thermoadaptation in an Ancestral Diterpene Cyclase by Altered Loop Stability. J. Phys. Chem. B. 2022, 126, 3809–3821. doi: 10.1021/acs.jpcb.1c10605

- B. Guo, S. Reddy Vanga, P. Saenz-Mendez, X. Lopez-Lorenzo, S. Rönnblad Ericsson, Y. Fang, X. Ye, K. Schriever, E. Bäckström, A. Biundo, R. A Zubarev, I. Furó, M. Hakkarainen, P.-O. Syrén*. Conformational selection in biocatalytic plastic degradation by PETase. ACS Catal. 2022, 12, 3397–3409. doi:10.1021/acscatal.1c05548


2021
- A. Stamm, J. Öhlin, C. Mosbech, P. Olsén, B. Guo, E. Söderberg, A. Biundo, L. Fogelström, S. Bhattacharyya, U. T. Bornscheuer, E. Malmström, P.-O. Syrén*. Pinene-based oxidative synthetic toolbox for scalable polyester synthesis. JACS Au 2021, 11, 1949–1960. doi: 10.1021/jacsau.1c00312d

- K. Schriever, P. Saenz-Mendez, R. Srilakshmi Rudraraju, N. M. Hendrikse, E. P. Hudson, A. Biundo, R. Schnell, P.-O. Syrén*. Engineering of Ancestor as a Tool To Elucidate Structure, Mechanism and Specificity of Extant Terpene Cyclase. J. Am. Chem. Soc. 2021, 143, 3794–3807. doi: 10.1021/jacs.0c10214

- C. Jönsson, R. Wei, A. Biundo, J. Landberg, L. Schwarz Bour, F. Pezzotti, A. Toca, L. M. Jacques, U. T. Bornscheuer, P.-O. Syrén*. Biocatalysis in the recycling landscape for synthetic polymers and plastics towards circular textiles. ChemSusChem 2021, 14, 4028–4040. https://doi.org/10.1002/cssc.202002666

- A. E. Alexakis, J. Engström, A. Stamm, A. V. Riazanova, C. J. Brett, S. V. Roth, P-O Syrén, L. Fogelström, M. S. Reid, E. Malmström. Modification of cellulose through physisorption of cationic bio-based nanolatexes – Comparing emulsion polymerization and RAFT-mediated polymerization-induced self-assembly. Green Chem. 2021,23, 2113.
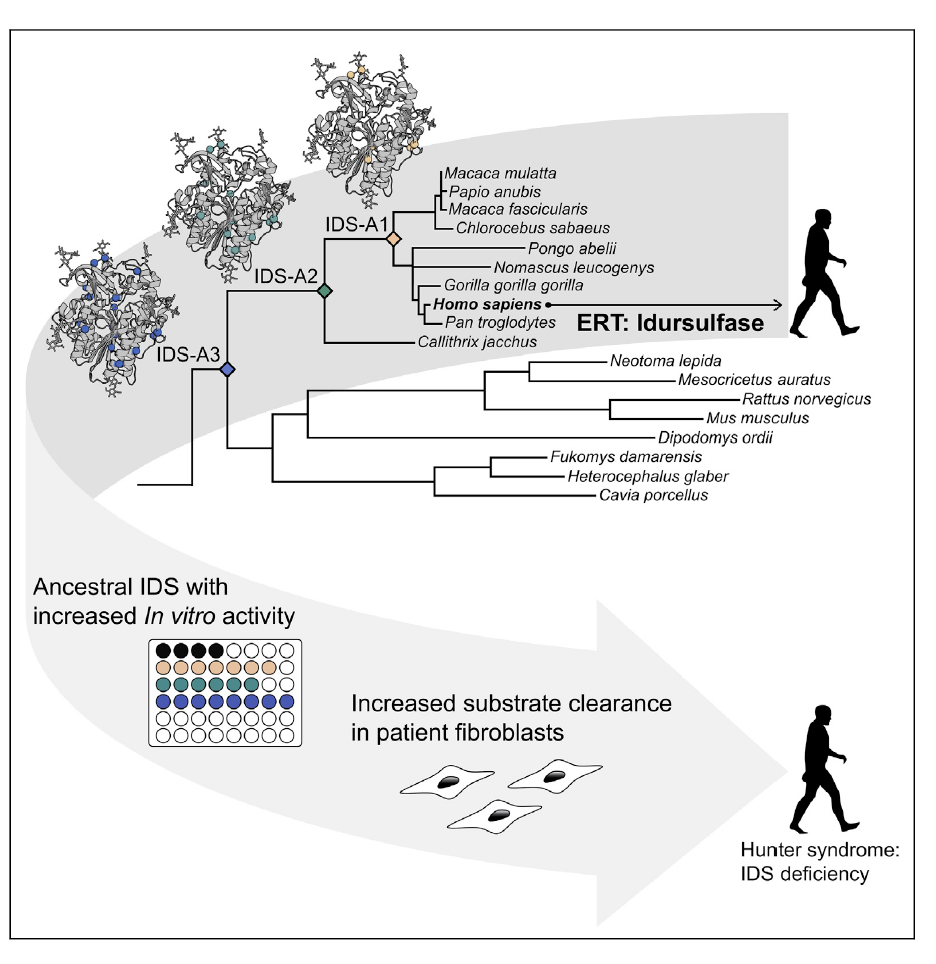
- N. M. Hendrikse, A. Sandegren, T. Andersson, J. Blomqvist, Å. Makower, D. Possner, C. Su, N. Thalén, A. Tjernberg, U. Westermark, J. Rockberg, S. Svensson Gelius, P.-O. Syrén*, E. Nordling. Ancestral lysosomal enzymes with increased activity harbor therapeutic potential for treatment of Hunter syndrome. iScience 2021, 24, 102154. https://doi.org/10.1016/j.isci.2021.102154
- A. Hunold, W. Escobedo-Hinojosa, E. Potoudis, D. Resende, T. Farr, P.-O. Syrén, B. Hauer. Assembly of a Rieske non-heme iron oxygenase multicomponent system from Phenylobacterium immobile E DSM 1986 enables pyrazon cis-dihydroxylation in E. coli. Appl. Microbiol. Biotechnol. 2021, 105, 2003–2015.
2020
- S. Zokaei, R. Kroon, J. Gladisch, B. Paulsen, A. I. Hofmann, G. Persson, A. Stamm, P.-O. Syrén, E. Olsson, J. Rivnay, E. Stavrinidou, A. Lund, C. Müller. Toughening of a Soft Polar Polythiophene through Copolymerization with Hard Urethane Segments. Adv. Sci. 2020, 2002778.
- I Biundo, A. Stamm, U. T. Bornscheuer, P.-O. Syrén*, Enzymatic synthesis of pinene-derived lactones. Applied Biocatalysis: The Chemist’s Enzyme Toolbox. 2020, pp 319-326. (First Edition, John Wiley & Sons Ltd).
- N. Hendrikse, A. Holmberg Larsson, S. Svensson Gelius, S. Kuprin, E. Nordling, Per-Olof Syrén*. Ancestral phenylalanine/tyrosine ammonia-lyases have potential for supplementary treatment to Nitisinone of hereditary tyrosinemia. Sci. Rep. 2020, 10, 1315.
- W. Farhat, A. Biundo, A. Stamm, E. Malmström, P.-O. Syrén*. Lactone monomers obtained by enzyme catalysis and their use in reversible thermoresponsive networks. J. Appl. Polym. Sci. 2020, 137, 48949.
2019
- Biundo, R. Subagia, M. Maurer, D. Ribitsch, P.-O. Syrén*, G. M. Guebitz. Switched reaction specificity in polyesterases towards amide bond hydrolysis by enzyme engineering. RSC Adv. 2019, 9, 36217-36226.
- Special issue on emerging future research leaders: Stamm, A. Biundo, B. Schmidt, J. Brücher, S. Lundmark, P. Olsén, L. Fogelström, E. Malmström, U. T. Bornscheuer, P.-O. Syrén* A retrobiosynthesis-based route to generate pinene-derived polyesters. ChemBioChem 2019, 20, 1664-1671.

- Special issue on Chemo-enzymatic cascade reactions: Farhat, A. Stamm, M. Robert-Monpate, A. Biundo, P.-O. Syrén*. Biocatalysis for terpene-based polymers. Z. Naturforsch. C. 2019, 74, 91–100.
- A. Stamm, M. Tengdelius, B. Schmidt, J. Engström, P. O. Syrén, L. Fogelström, E. Malmström. Chemo-enzymatic pathways toward pinene-based renewable materials. Green Chem. 2019, 21, 2720–2731.

2018
- M. Hendrikse, G. Charpentier, E. Nordling, P.-O. Syrén*, Ancestral diterpene cyclases show increased thermostability and substrate acceptance. FEBS J 2018, 285 4660–4673.

- Elected among the top 25 papers in JOC during 2018 Featured on the cover of J. Org. Chem. P.-O. Syrén*, Enzymatic hydrolysis of tertiary amide bonds by anti nucleophilic attack and protonation. J. Org. Chem. 2018, 83, 13543–13548.

2016-2017
- A. Eriksson, C. Kürten, P.-O. Syrén*, Protonation-initiated cyclization by a class II terpene cyclase assisted by tunneling. ChemBioChem 2017, 18, 2301-2305.
- A. Eriksson, C. Kürten, P.-O. Syrén*, Cover Feature: Protonation-initiated cyclization by a class II terpene cyclase assisted by tunneling. ChemBioChem 2017, 18, 2293.
- J. Fagerland, D. Pappalardo, B. Schmidt, P.-O. Syrén, A. F. Wistrand, Template assisted enzymatic synthesis of oligopeptides from a polylactide chain. Biomacromolecules, 2017, 18, 4271−4280.
- C. Gustafsson, S. Vassiliev, C. Kürten, P.-O. Syrén, T. Brinck. MD Simulations Reveal Complex Water Paths in Squalene–Hopene Cyclase: Tunnel-Obstructing Mutations Increase the Flow of Water in the Active Site. ACS Omega 2017, 2, 8495−8506.
- M. J. Fink, P.-O. Syrén*, Redesign of water networks for efficient biocatalysis. Curr. Opin. Chem. Biol. 2017, 37, 107-114.
- S. C. Hammer, P.-O. Syrén#, B. Hauer. Substrate Pre-Folding and Water Molecule Organization Matters for Terpene Cyclase Catalyzed Conversion of Unnatural Substrates. ChemistrySelect, 2016, 1, 3589–3593.
- P.-O. Syrén, S. Henche, A. Eichler, B. Nestl, B. Hauer. Squalene-hopene cyclases – evolution, dynamics and catalytic scope. Curr. Opin. Struct. Biol. 2016, 41, 73-82.
- Special issue: Enzyme catalysis: C. Kürten, B. Carlberg, P.-O. Syrén*. Mechanism-guided discovery of an esterase scaffold with promiscuous amidase activity. Catalysts, 2016, 6, 90.
- C. Kürten, P.-O. Syrén*. Unraveling entropic rate acceleration induced by solvent dynamics in membrane enzymes. J. Vis. Exp. 2016, 107, e53168, doi:10.3791/53168.
Publications before 2016
- C. Kürten, M. Uhlén, P.-O. Syrén*. Overexpression of functional human oxidosqualene cyclase in Escherichia coli. Protein Express Purif, 2015, 115, 46-53.
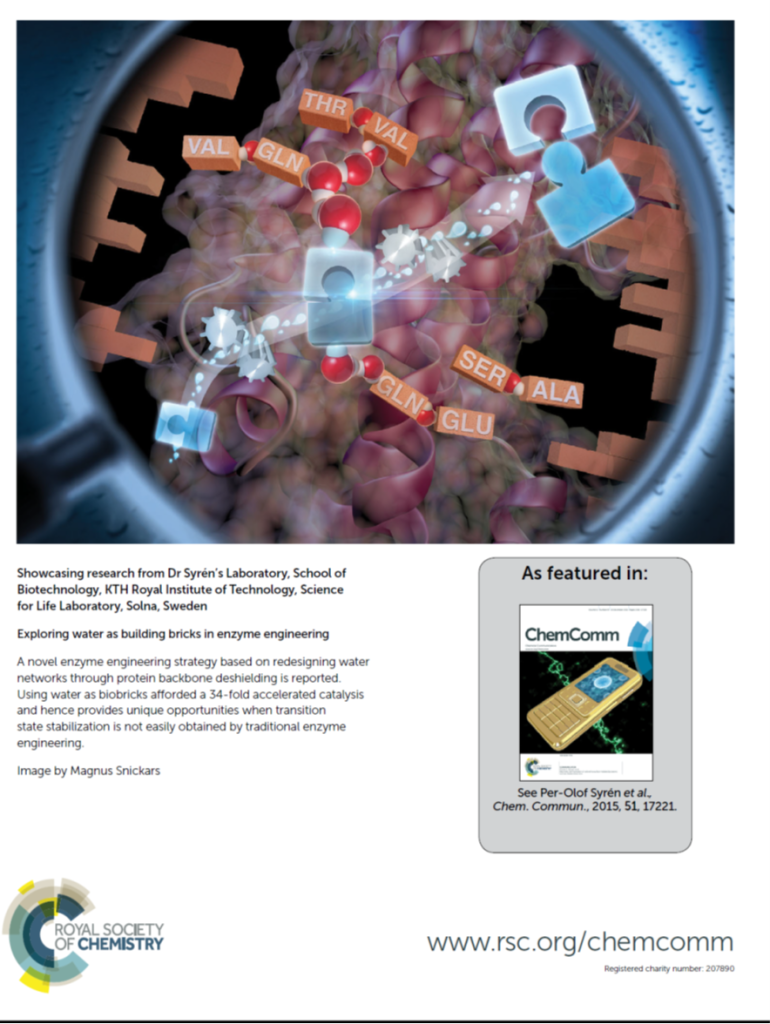
- Featured on the cover of Chemical Communications: P. Hendil-Forssell, M. Martinell, P.-O. Syrén*. Exploring water as building bricks in enzyme engineering. Chem. Commun. 2015, 51, 17221-17224.
- P.-O. Syrén*, S.C. Hammer, B. Claasen, B. Hauer. Entropy is Key to the Formation of Pentacyclic Terpenoids by Enzyme-Catalyzed Polycyclization. Angew. Chem. Int. Ed. 2014, 126, 4945-4949.
- P.-O. Syrén*, The solution to nitrogen inversion in amidases. FEBS J 2013, 280, 3069-3083.
- M. Seitz, P.-O. Syrén, L. Steiner, J. Klebensberger, B. Nestl, B. Hauer, Synthesis of heterocyclic terpenoids by promiscuous squalene-hopene cyclases. ChemBioChem 2013, 14, 436-439.
- S. C. Hammer#, P.-O. Syrén#, M. Seitz, B. Nestl, B. Hauer, Squalene hopene cyclases: highly promiscuous and evolvable catalysts for stereoselective C-C and C-X bond formation. Curr. Opin. Chem. Biol. 2013, 17, 293-300.
- P.-O. Syrén*, F. Le Joubioux, Y. Ben Henda, T. Maugard, K. Hult, M. Graber, Proton shuttle mechanism in the transition state of lipase catalyzed N-acylation of amino alcohols. ChemCatChem 2013, 5, 1842-1853.
- S. C. Hammer, J. Dominicus, P.-O. Syrén, B. Nestl, B. Hauer, Stereoselective Friedel–Crafts alkylation catalyzed by squalene hopene cyclases. Tetrahedron, 2012, 68, 7624-7629.
- P.-O. Syrén, P. Hendil-Forssell, L. Aumailley, W. Besenmatter, F. Gounine, A. Svendsen, M. Martinelle, K. Hult, Esterases with an introduced amidase-like hydrogen bond in transition state display increased amidase specificity. ChemBioChem 2012, 13, 645–648.
- P.-O. Syrén, K. Hult, Amidases have a hydrogen bond that facilitates nitrogen inversion but esterases have not. ChemCatChem 2011, 3, 853-860.
- K. Engström, M. Vallin, P.-O. Syrén, K. Hult, J.-E. Bäckvall, Mutated variant of Candida antarctica lipase B in (S)-selective dynamic kinetic resolution of secondary alcohols. Org. Biomol. Chem. 2011, 9, 81-82.
- P.-O. Syrén, K. Hult, Substrate Conformations Set the Rate of Enzymatic Acrylation by Lipases. ChemBioChem 2010, 11, 802 – 810.
- S. Marton, V. Léonard-Nevers, P.-O. Syrén, C. Bauer, S. Lamare, K. Hult, V. Tranc, M. Graber, Mutations in the stereospecificity pocket and at the entrance of the active site of Candida antarctica lipase B enhancing enzyme enantioselectivity. J. Mol. Catal. B: Enzym. 2010, 65, 11-17.
- P.-O. Syrén, E. Lindgren, H. W. Hoeffken, C. Branneby, S. Maurer, B. Hauer, K. Hult, Increased activity of enzymatic transacylation of acrylates through rational design of lipases. J. Mol. Catal. B: Enzym. 2010, 65, 3-10.
- M. Vallin, P.-O. Syrén, K. Hult, Mutant Lipase-Catalyzed Kinetic Resolution of Bulky Phenyl Alkyl sec-Alcohols: A Thermodynamic Analysis of Enantioselectivity. ChemBioChem 2010, 11, 411-416.
- P.-O. Syrén, A. Rozkov, S. R. Schmidt, P. Strömberg, Milligram scale parallel purification of plasmid DNA using anion-exchange membrane capsules and a multi-channel peristaltic pump. J. Chromatogr. B 2007, 856, 68-74.
- I. V. Pavlidis, N.M. Hendrikse, P.-O. Syrén*, Computational Techniques for Efficient Biocatalysis. Modern Biocatalysis: Advances Towards Synthetic Biological Systems. 2018, 5, 119-152 (Royal Society of Chemistry).
- P.-O. Syrén*, Understanding esterase and amidase reaction specificities by molecular modelling. Understanding enzymes; Function, Design, Engineering and Analysis. 2016, 15, 523-558 (PanStandford Publishing).